Assay points.
-
ALSU-TCSF1R-A500
500
-
ALSU-TCSF1R-A10K
10,000
-
ALSU-TCSF1R-A50K
50,000
-
ALSU-TCSF1R-A-HV
100
Assay Principle
The AlphaLISA™ SureFire® Ultra™ assay enables the rapid and sensitive detection of total and phosphorylated cellular proteins. AlphaLISA™ assays utilize two bead types: Acceptor Beads and Donor Beads. The Acceptor Bead is coated with a proprietary CaptSure™ agent to specifically immobilize the assay specific antibody which is labeled with a CaptSure™ tag. The Donor Bead is coated with streptavidin to capture the biotinylated antibody.
In the presence of a target protein, the two target-specific antibodies bring Donor and Acceptor Beads into close proximity. When the Donor Beads are activated by a laser (680 nm), singlet oxygen is transferred to the Acceptor Bead leading to the production of an Alpha signal. The amount of light emission (615 nm) from the Acceptor Bead is directly proportional to the amount of target protein present in the sample. If an Acceptor Bead is not in close proximity (i.e. 200 nm) of a Donor bead, little to no signal is produced over background.
The assay can be executed in a 1-plate or 2-plate assay protocol (Refer to Manual for more details).
1-plate assay protocol: culturing of cells, treatment, lysis and assay are performed in a single well, enabling miniaturization in high throughput screening programs.
2-plate assay protocol: cells are cultured and treated in a 96-well culture plate and lysates are then transferred into a separate plate for assay. This format allows the evaluation of multiple targets from a single lysate.
Technical Specific Data
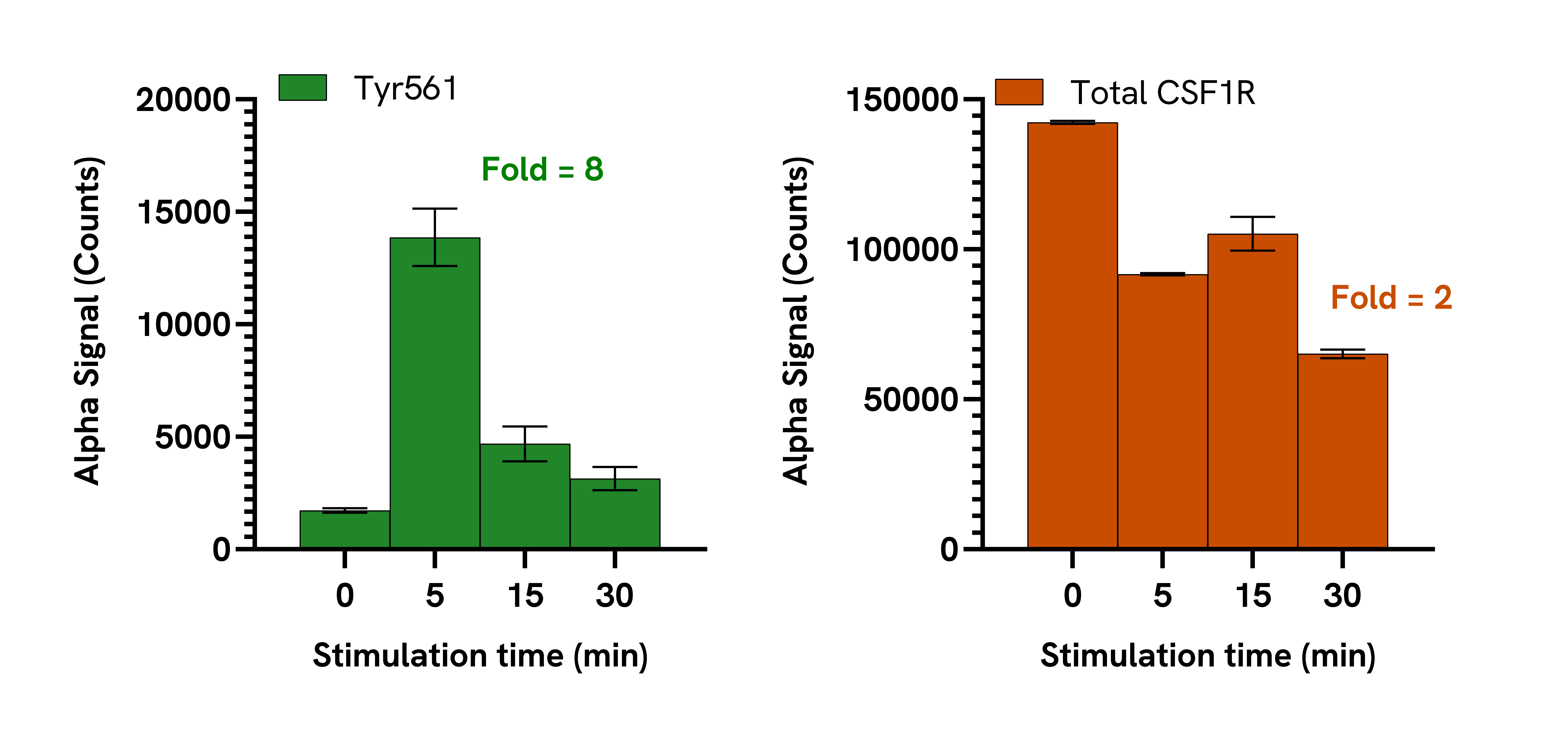
Data obtained with a 2-plate, 2-incubation protocol. PBMCs were isolated from healthy donors and cultured for 7 days in complete DMEM containing 20 ng/mL M-CSF to differentiate them into macrophages. Macrophages were seeded in a 96-well plate (40,000 cells/well) in complete DMEM and incubated overnight. The cells were starved for 2 hours in HBSS + 0.1% BSA and then treated with 100 ng/mL M-CSF for the indicated time points. Cells were then lysed with Lysis Buffer and assayed separately for Phospho (Tyr561) and Total CSF1R using respective SureFire® Ultra™ kits. Equivalent to approximately 4,000 cells/datapoint.
Data obtained with a 2-plate, 2-incubation protocol. BeWo cells were seeded at 60,000 cells/well in a 96 well plate and incubated overnight. The cells were starved for 2 hours in HBSS + 0.1% BSA and then treated with M-CSF at the indicated concentrations for 5 minutes. Cells were then lysed with Lysis Buffer and assayed separately for Phospho (Tyr561) and Total CSF1R using respective SureFire® Ultra™ kits. Equivalent to approximately 6,000 cells/datapoint.
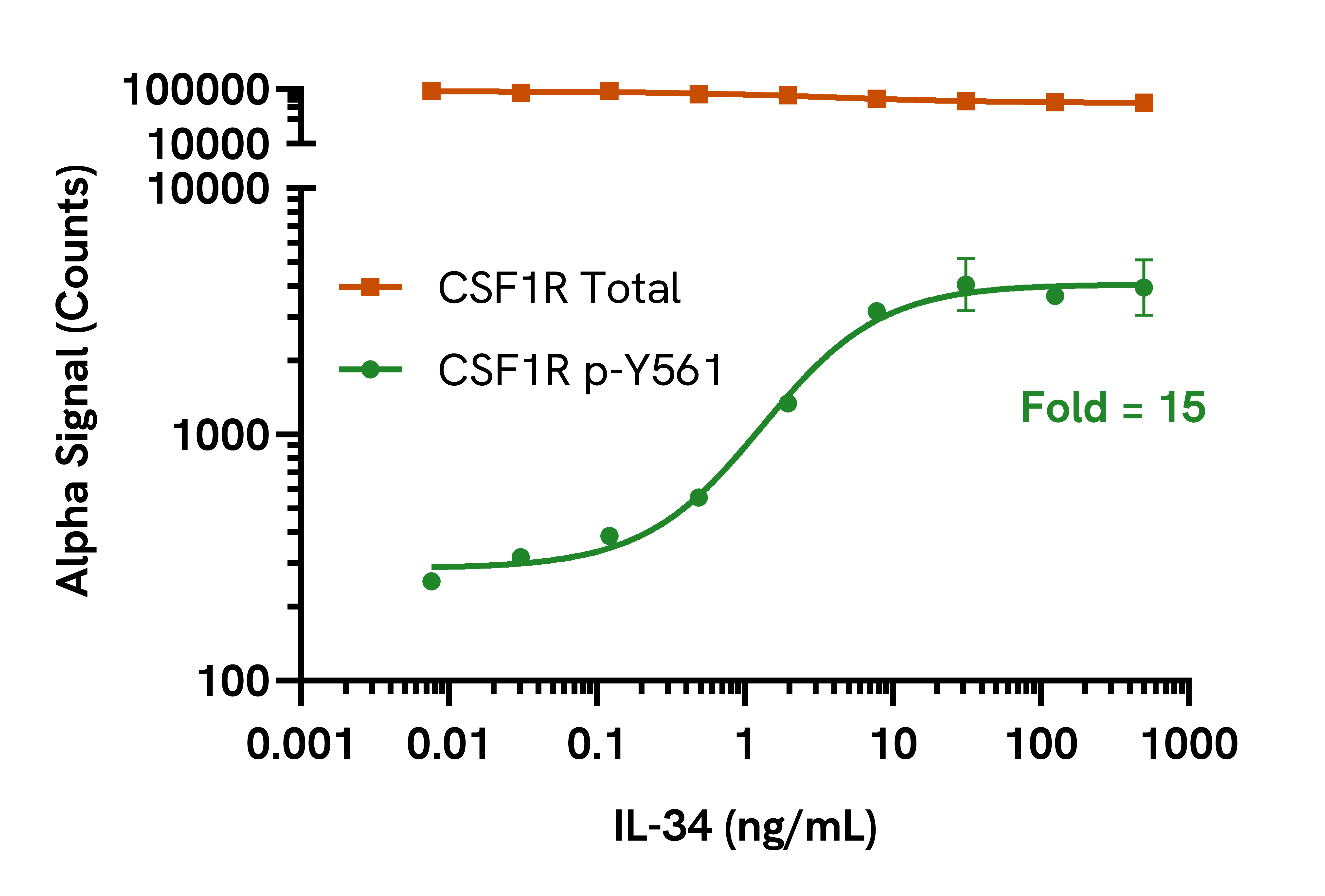
Data obtained with a 2-plate, 2-incubation protocol. THP-1 cells were seeded at 200,000 cells/well in HBSS + 0.1% BSA and starved for 2 hours. Cells were treated with IL-34 at the indicated concentrations for 5 minutes. Cells were spun down and lysed in Lysis Buffer and assayed separately for Phospho (Tyr561) and Total CSF1R using respective SureFire® Ultra™ kits. Equivalent to approximately 20,000 cells/datapoint.
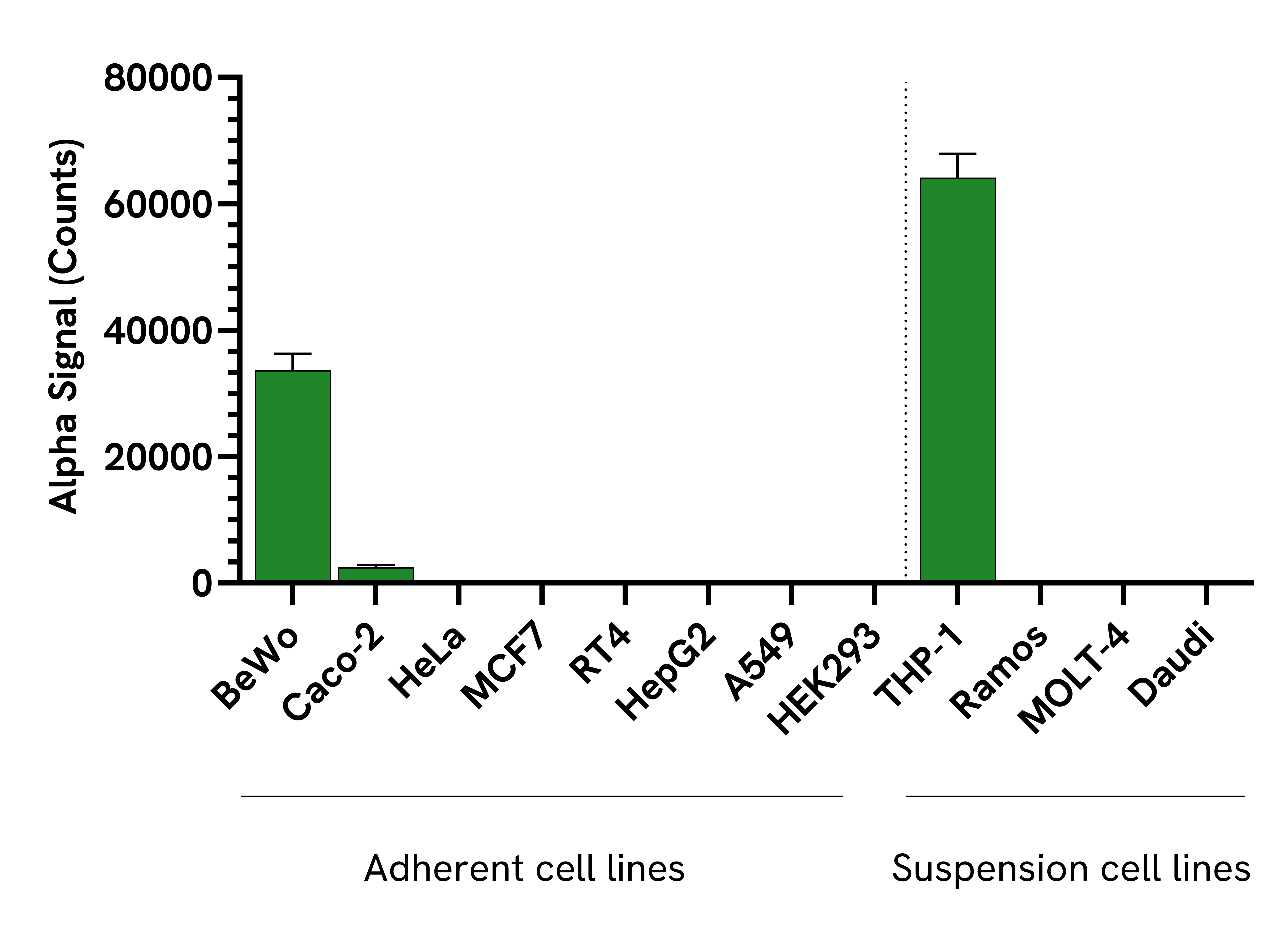
Data obtained from measurement of Total CSF1R protein levels in various cell types. Adherent cell lines were seeded at 40K cells/well and incubated overnight and lysed with Lysis Buffer. Suspension cell lines were seeded at 400K cells/well and lysed with Lysis Buffer. Suspension and adherent cell lysates were then assayed for Total CSF1R using the SureFire® Ultra® kit. Equivalent to approximately 2,000 cells/datapoint for adherent and 20,000 cells/datapoint for suspension cell lines.
Manuals & downloads.
-
PDF
-
PDF
Detection Kit Manual
-
Tech Spec
Technical Data Sheet
Safety Data Sheets
-
Safety Data Sheet (SDS)
Safety Data Sheet EU-EN
-
Safety Data Sheet (SDS)
Safety Data Sheet US